Infobox drug/testcases-FDA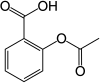 | 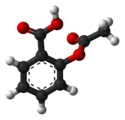 | |
| Pronunciation | acetylsalicylic acid /əˌsiːtəlˌsælᵻˈsɪlᵻk/ |
|---|
| Trade names | Bayer Aspirin, others |
|---|
| Other names | - 2-acetoxybenzoic acid
- o-acetylsalicylic acid
- acetylsalicylic acid
- acetyl salicylate
|
|---|
| AHFS/Drugs.com | Monograph |
|---|
| MedlinePlus | a682878 |
|---|
| License data |
|
|---|
Pregnancy
category | |
|---|
Routes of
administration | By mouth, rectal |
|---|
| ATC code | |
|---|
|
| Legal status |
|
|---|
|
| Bioavailability | 80–100%[8] |
|---|
| Protein binding | 80–90%[9] |
|---|
| Metabolism | Liver (CYP2C19 and possibly CYP3A), some is also hydrolysed to salicylate in the gut wall.[9] |
|---|
| Elimination half-life | Dose-dependent; 2–3 h for low doses (100 mg or less), 15–30 h for large doses.[9] |
|---|
| Excretion | Urine (80–100%), sweat, saliva, feces[8] |
|---|
|
2-acetyloxybenzoic acid [10]
| | CAS Number | |
|---|
| PubChem CID | |
|---|
| IUPHAR/BPS | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEBI | |
|---|
| ChEMBL | |
|---|
| PDB ligand | |
|---|
|
| Formula | C9H8O4 |
|---|
| Molar mass | 180.16 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
| Density | 1.40 g/cm3 |
|---|
| Melting point | 136 °C (277 °F) [5] |
|---|
| Boiling point | 140 °C (284 °F) (decomposes) |
|---|
| Solubility in water | 3 g/L |
|---|
|
InChI=1S/C9H8O4/c1-6(10)13-8-5-3-2-4-7(8)9(11)12/h2-5H,1H3,(H,11,12)  Y YKey:BSYNRYMUTXBXSQ-UHFFFAOYSA-N  Y Y
| | (verify) | |
Infobox drug/testcases-FDA |  | |
| Pronunciation | acetylsalicylic acid /əˌsiːtəlˌsælᵻˈsɪlᵻk/ |
|---|
| Trade names | Bayer Aspirin, others |
|---|
| Other names | - 2-acetoxybenzoic acid
- o-acetylsalicylic acid
- acetylsalicylic acid
- acetyl salicylate
|
|---|
| AHFS/Drugs.com | Monograph |
|---|
| MedlinePlus | a682878 |
|---|
| License data |
|
|---|
Pregnancy
category | |
|---|
Routes of
administration | By mouth, rectal |
|---|
| ATC code | |
|---|
|
| Legal status |
|
|---|
|
| Bioavailability | 80–100%[8] |
|---|
| Protein binding | 80–90%[9] |
|---|
| Metabolism | Liver (CYP2C19 and possibly CYP3A), some is also hydrolysed to salicylate in the gut wall.[9] |
|---|
| Elimination half-life | Dose-dependent; 2–3 h for low doses (100 mg or less), 15–30 h for large doses.[9] |
|---|
| Excretion | Urine (80–100%), sweat, saliva, feces[8] |
|---|
|
2-acetyloxybenzoic acid [10]
| | CAS Number | |
|---|
| PubChem CID | |
|---|
| IUPHAR/BPS | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEBI | |
|---|
| ChEMBL | |
|---|
| PDB ligand | |
|---|
|
| Formula | C9H8O4 |
|---|
| Molar mass | 180.16 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
| Density | 1.40 g/cm3 |
|---|
| Melting point | 136 °C (277 °F) [5] |
|---|
| Boiling point | 140 °C (284 °F) (decomposes) |
|---|
| Solubility in water | 3 g/L |
|---|
|
InChI=1S/C9H8O4/c1-6(10)13-8-5-3-2-4-7(8)9(11)12/h2-5H,1H3,(H,11,12)  Y YKey:BSYNRYMUTXBXSQ-UHFFFAOYSA-N  Y Y
| | (verify) | |

